Unforeseen aspects of expression patterns of terminal differentiation genes that we discovered over the years have drawn us into areas of neurobiology that go beyond development and provided us with insights about how the environment interacts with individual neurons and the nervous system as a whole:
(a) How neurons perceive environmental taste cues
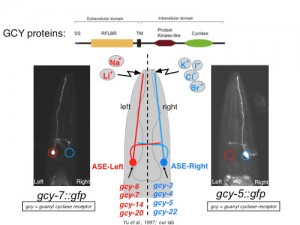
The left/right asymmetric expression of receptor-type guanylyl cyclases (rGCs) in the ASE(L/R) salt receptor neurons, as well as the generally poorly understood mechanisms of salt detection in any system ignited our interest in studying rGCY function (Figure 1). We have found that individual rGCYs are required for the processing of specific taste cues [1]. Domain swap experiments demonstrated that their extracellular domain is required for specific sensory responses, supporting the view that they may work as salt receptors (unpubl.data).
(b) How neuronal anatomy and circuitry is modulated by environmental conditions
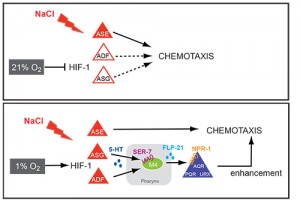
Our analysis of expression patterns of terminal differentiation features of serotonergic neurons has led us to discover that environmental oxygen levels (i) impact on the migration and axon pathfinding of serotonergic neurons[2] and (ii) induce a rerouting of chemosensory information through a novel, serotonin- and neuropeptide-mediated circuit to ensure the robustness of the gustatory response under adverse conditions[3](Figure 2). We are currently investigating how an enviromentally and hormonally controlled altered life state of the worm, the dauer state, impacts on the execution of terminal differentiation programs in the nervous system.
(c) How neuronal identity and circuitry is affected by sex
The recent elucidation of the connectome of the male by the Emmons lab (www.wormwiring.com) has prompted us (a) to ask how differences in male and hermaphrodite neuronal circuitry are genetically programmed and (b) to investigate whether there are other molecular distinction in terminal differentiation programs in male and hermaphroditic neurons.
(d) Facing environmental challenges: How the anatomy of the nervous system is maintained
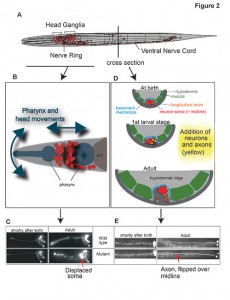
Another gfp-based expression survey of terminal differentiation genes had led us to identify an unusual family of small, secreted Ig domain proteins (ZIG proteins), required to maintain the overall structural organization of the nervous system by counteracting environmental forces (generated by movement of the animal) that mechanically dislocate neuronal structures[4-7](Figure 3). These findings prompted us to search for more molecules with roles in maintaining nervous system architecture, leading us to the identification of maintenance roles of the FGF receptor egl-15 [8], the IgCAM sax-7 [9] and the extracellular matrix protein dig-1 [10].